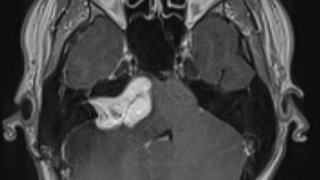
UT Southwestern is one of just four medical centers nationwide selected by the National Institute of Mental Health to conduct the EMBARC study, which hopes to identify biosignatures that are linked to effective treatments for depression. UTSW is currently seeking people to participate in the study.
“Today, if you go to your doctor about depression, he or she has over 20 different medications and psychotherapies available to help, but it’s a guessing game to figure out which one to try, and you may have to try several until you find one that’s right for you,” says Madhukar Trivedi, M.D., Professor of Psychiatry and Lead Principal Investigator of the study.
Biosignatures could potentially change that treatment model.
“A biosignature is a unique combination of features about you and your illness,” Dr. Trivedi says. “If you see a doctor for a sore throat, he or she checks your symptoms and combines that information with medical tests that identify the specific germ making you sick. Then the physician chooses the right treatment. We think the same approach will work for depression, but we need help, through this study, to learn which tests provide the best answers. With a biosignature, doctors will be able to treat patients with the medication that has the best chance of success first.”

What’s Involved In The Study
Brain scans, blood draws, and other noninvasive tests. Participants will be given Food and Drug Administration-approved treatments at no cost to see if symptoms improve. Participants are also eligible for compensation.
The EMBARC Study is no longer recruiting patients.