Using magnets to treat major depressive disorder: Clinical trial shows promise
May 11, 2022
UT Southwestern has reached the midpoint of a clinical trial to see whether magnetic seizure therapy (MST) – a novel option that uses magnets to stimulate the brain to treat medication-resistant depressive disorder (MDD) – is as effective as electroconvulsive therapy (ECT).
The CREST-MST trial, funded by the National Institute of Mental Health, at this time is available to U.S. patients only at UT Southwestern. Together with a center in Toronto, Canada, we are currently the only two North American academic medical centers offering MST to treat major depressive disorder.
Clinical data show that, for reasons we are still investigating, seizure-related changes in brain chemistry can improve or reverse MDD symptoms. Electroconvulsive therapy induces therapeutic seizures in a patient’s brain using electrical currents and is one of the most efficacious antidepressant treatments.
Approximately 80% of patients with severe MDD respond to ECT, compared with just 50% who respond to medication. While ECT is highly effective, some patients are understandably concerned about potential side effects, such as short-term memory loss.
Data from the CREST-MST trial will help us determine whether MST, which carries fewer cognitive side effects, might be an effective alternative. We hope the trial will provide the necessary data needed for the U.S. Food and Drug Administration to approve MST as a new safe and effective antidepressant therapy.
How does magnetic seizure therapy work?
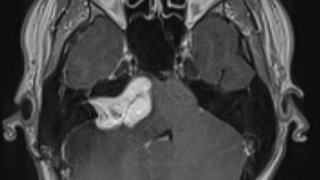
In a clinical setting, a specialist trained in MST uses a magnetic coil – pre-measured to fit comfortably – to trigger therapeutic seizures in a patient’s brain. The seizure activity increases blood flow to the areas where depression originates, which might aid in depression symptom reduction.

Compared with the electrical currents used in ECT, magnetic currents do not penetrate as deeply and can be focused more precisely, which may result in a relatively better cognitive safety profile. Both ECT and MST are performed when a patient is under general anesthesia. The controlled seizures typically last only 30 to 60 seconds, and total treatment time per session is approximately 15 minutes, followed by recovery from anesthesia.
The CREST-MST trial is a randomized, double-blind study, meaning participants do not know which treatment they are getting. Patients are randomly assigned to receive either ECT or MST. Treatment is given three days a week for a maximum of 21 treatment sessions.
Even if MST is shown to be as effective as ECT, it is unlikely we will stop prescribing ECT for certain patients. Clinical experience has demonstrated that bilateral ECT remains superior to unilateral ECT and MST in terms of clinical efficacy. And though bilateral ECT carries more cognitive adverse effects, this option remains available through our clinical program at UT Southwestern for people who do not have an optimal response to the study intervention.
Who is eligible for the CREST-MST trial?
We are enrolling approximately 130 patients, age 18 and older who have been diagnosed with unipolar MDD. Adults with significant neurological or medical conditions and patients who are pregnant are not eligible. See the full trial eligibility criteria.
Before starting ECT or MST, we will work to optimize participants’ antidepressant medication plan, and patients will continue taking their medications during the clinical trial. Most participants pause psychotherapy during the treatment series, though it is strongly encouraged to resume psychotherapy after study treatments conclude to support ongoing mental health and wellness.
The future of interventional psychiatry
UT Southwestern’s psychiatry experts offer all available FDA-approved interventional treatments for major depressive disorder, as well as some advanced clinical trial therapies that are not yet widely available.

Along with the MST trial, we’re engaged in two deep brain stimulation (DBS) studies for MDD with Nader Pouratian, M.D., Ph.D., Chair of Neurosurgery at UT Southwestern. We are also conducting DBS for obsessive compulsive disorder (OCD) clinically.
Unfortunately, advanced therapies such as ECT have been plagued by misinformation and negative stigma throughout the years – namely, ECT has been likened to the outdated “treatment” in the 1975 movie One Flew Over the Cuckoo’s Nest. While that stigma is lifting, we still have work to do.
Through ongoing research to expand our clinical services, we offer hope and healing to patients coping with severe and medication-resistant depression – patients who once had limited or no remaining treatment options. It would be remarkable to add MST as another modality that could potentially provide relief or remission of severe major depressive disorder symptoms for our patients.
To visit with a psychiatrist about MDD treatment options, call 214-645-8300 or request an appointment online.