When a patient is diagnosed with a malignant brain tumor, also known as glioblastoma, it’s a life-altering event. Time is of the essence, and early and expert brain tumor surgery can make a significant difference in the patient’s outcome.
At UT Southwestern, our brain tumor team uses cutting-edge advanced technology to make it easier to see and remove a patient’s entire tumor. In 2019, we performed the first fluorescence-guided brain tumor surgeries in the Dallas-Fort Worth region and have continued to expand the use of this technique to eligible patients since then.
Studies have shown that the treatment, which uses an optical imaging agent called Gleolan to help visualize tumor cells during surgery, gives patients a better chance for long-term survival and extends the time it takes brain cancer cells to return.
UT Southwestern’s Peter O’Donnell Jr. Brain Institute is one of a select few institutions across the country that is able to offer Gleolan to patients, and in our extensive experience so far, we expect it will quickly become a vital tool in our quest to provide glioblastoma patients with the best possible options and outcomes.
Techniques to tackle brain tumors
Glioblastoma is the most common malignant brain tumor in the U.S. About 12,000 people each year are diagnosed with it. (In 2018, Arizona Sen. John McCain died from a glioblastoma. Nine years to the day earlier, McCain’s colleague and friend Sen. Edward Kennedy of Massachusetts died of the same aggressive form of brain cancer.)
Glioblastoma is also one of the most difficult cancers to treat in the human body. The tumors typically are not contained in one specific mass and they can have finger-like vines that extend into nearby areas of the brain.
The standard of care for glioblastoma is early surgical resection, or removal, of the tumor, followed by chemotherapy and radiation. But the effectiveness of the surgery can be limited by several factors, including visibility and position of the tumor.
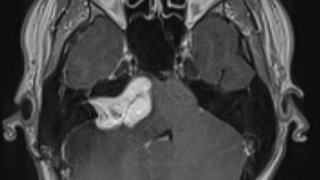
Under a white light, it can be difficult to distinguish between a glioblastoma and normal brain tissue. If the tumor is in an area of the brain that includes critical functions, like language and memory, that can make it even more challenging to remove.
'Gleolan (aminolevulinic acid hydrochloride) comes in liquid form and patients drink it two to four hours before surgery. The tumor glows under a blue light microscope, making it easier for the surgeon to differentiate the glioblastoma from normal brain tissue.'
Kalil Abdullah, M.D.

Essentially, all patients have some recurrence of their tumor. Even with advanced surgical resection techniques, microscopic cancer cells are left behind because tumor cells infiltrate surrounding brain tissue. That is why surgery is followed by chemotherapy and radiation.
But we now know that our ability to remove as much of the tumor as possible is directly tied to long-term patient survival. Over the last several decades, neurosurgeons have developed techniques to remove as much tumor as is safely possible. These include:
- Intraoperative neuro-navigation, which is similar to a real-time GPS system for the brain
- Intraoperative motor mapping, which allows neurosurgeons to stimulate the brain while the patient is under anesthesia
- Awake craniotomies, which make it possible to talk to patients, have them take quizzes, even play a musical instrument during surgery so we can achieve resection and preserve vital areas of the brain. (We numb the scalp with lidocaine to ensure the patient is pain-free during the procedure.)
These techniques have all helped neurosurgeons push the boundaries of tumor removal while keeping patients safe. Gleolan takes these medical advances one step further.
A drink that lights up tumors
Gleolan (aminolevulinic acid hydrochloride) was first introduced in Europe and has been approved there for several years. It comes in liquid form and patients drink it two to four hours before surgery. It then circulates through the body until it reaches the tumor, where it’s metabolized and synthesized selectively by malignant tumor cells.
Surgeons use a highly specific “blue light” microscope and filter, which allows the tumor to fluoresce and “glow” a bright orange color, helping them differentiate between normal brain tissue and tumor cells. It can also help neurosurgeons reach any remaining tumor cells that might have been left behind.
In randomized, controlled trials, Gleolan was shown to increase the number of patients who had complete removal of their malignant brain tumors by 26%.
The U.S. Food and Drug Administration gave Gleolan the green light in mid-2017, and it has slowly made its way to select major health care centers across the U.S.
Several factors contributed to the meticulous rollout:
- First, it requires a highly advanced microscope and filter to see the fluorescence.
- Second, a hospital must see and treat enough brain tumors so that it has systems in place to select which patients will benefit from Gleolan-assisted removal.
- Finally, surgeons must have experience in malignant brain tumor surgery, and experience using the drug.
Our team is highly experienced and has been performing resections using Gleolan on numerous patients since fall 2019. In many of these patients, Gleolan helped us to achieve excellent results in highly delicate regions of the brain that control movement, speech, and consciousness. In several cases, Gleolan was used when a glioblastoma needed to be removed from a patient while they were awake.
(Gleolan can be especially helpful when patients are awake during brain surgery, because the more efficient the procedure is, the better.)
In all cases, Gleolan made it easier to use a smaller brain incision to get access to the tumor, and also allowed our team to remove as much of the tumor as possible.
Low risk, high reward
While Gleolan is relatively new to the U.S., I suspect it will become a standard of care for treating glioblastoma in the next five years. Why?
It presents almost no risk to the patient. The only reported side effects have been moderate gastrointestinal discomfort and sensitivity to light. We ask patients to stay in a dimly lit environment for one day after surgery to guard against skin sensitivity.
It is also a surprisingly cost-effective treatment when compared with other medical and surgical techniques, according to a recently published study.
The treatment of glioblastoma starts with successful neurosurgery, but we also work closely with our colleagues in the Harold C. Simmons Comprehensive Cancer Center and in radiation oncology to provide multidisciplinary, comprehensive care for our patients.
We may not yet have a cure for glioblastoma, but as a comprehensive brain tumor center and the only National Cancer Institute-designated Comprehensive Cancer Center in North Texas, UT Southwestern is continually striving to give our patients the best possible chance for survival.
If you or a loved one is dealing with glioblastoma, you can request an appointment online or call 214-645-8300.

Our Progress
Learn about other breakthroughs happening at the O’Donnell Brain Institute.