March 28, 2017, was an important day for patients who have multiple sclerosis (Multiple Sclerosis). On that day, the drug Ocrevus (OH-kreh-vus) became the first treatment ever approved by the Food and Drug Administration (FDA) for primary-progressive MS, the most aggressive form of the disease. Ocrevus, whose generic name is ocrelizumab, also is approved to treat patients who have relapsing-remitting MS.
A team of researchers led by Dr. Stephen Hauser at the University of California, San Francisco, spearheaded the research efforts on this medication and its predecessor, rituximab. I was a part of Dr. Hauser’s MS group for six years while I was at UCSF and participated in the original scientific efforts related to this class of medications.
After four decades of scientific study, patients in North Texas and nationwide will now be able to see the benefits of Ocrevus in their treatment plans. This medication is a huge advancement in the treatment of MS because of the improvements patients have shown while they take it. In fact, for patients with primary-progressive MS, Ocrevus is now the drug doctors should try first in the treatment process.
It’s important to understand what Ocrevus does and what patients can expect from treatment. Talk to your neurologist or MS specialist about whether you should start receiving Ocrevus therapy.
How does Ocrevus work for MS?
MS is an autoimmune disorder. That means patients are injured by their own immune system. A patient with MS is exposed to some unknown trigger that excites the immune system. It could be a virus or something in the air or something the patient eats or drinks.
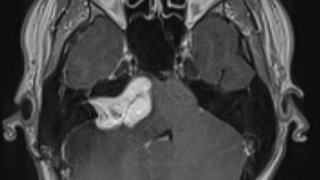
The trigger causes the cells of the immune system to attack the central nervous system and create lesions (damaged tissue) on the brain, spinal cord, and optic nerve, which is instrumental to sight. The body’s immune system includes T-cells and B-cells that normally help protect us from disease and infection, but these cells malfunction in people with MS.
For many years, researchers believed only T-cells were involved in the MS disease process; thus, many of the medications that have been available to treat MS target only T-cells. But the research into Ocrevus revealed that patients with MS also may have B-cells that are malfunctioning.
Ocrevus, a monoclonal antibody, targets and destroys B-cells with a certain kind of receptor (a molecule that lets the cells receive chemical signals from elsewhere in the body). The receptor that Ocrevus looks for is called CD20. Once it finds that receptor, it binds to that cell and pops it. The body then removes that destroyed cell from circulation.
Ocrevus is available from our Clinical Center for Multiple Sclerosis and other MS specialty centers. We see more than 5,000 patients with MS each year, and each patient receives a personalized treatment plan. We’re currently scheduling patients to start receiving Ocrevus infusions at our University Hospital Infusion Center – Aston. Patients will receive Ocrevus every six months. Call 214-645-8300 or request an appointment with our online form.
What are the benefits of Ocrevus?
In clinical trials, a significant number of patients given Ocrevus experienced higher levels of function and decreased levels of disability.
In patients with primary-progressive MS, studies showed a lower percentage of patients whose MS worsened over time – a distinguishing feature of the primary-progressive form of the disease. Researchers also saw that Ocrevus prevented new lesions in patients who were given the medication.
Ocrevus has a distinct advantage over its predecessor, rituximab, in that it’s made mostly of human proteins. Rituximab is made from a combination of human and mouse proteins. Some patients who take rituximab develop an immune response to the medication because their immune system senses the mouse proteins as foreign. Having no parts mouse, Ocrevus has a lower chance of the body’s immune system rejecting the medication.
One of the most surprising things to me is the low cost of the drug compared to the other therapies we have available. My hope is that the lower cost means patients can have greater access to the medication.
Here’s how the wholesale cost of Ocrevus compares to some of the most widely used medications in MS treatment:
- 25 percent annual savings compared to Betaseron
- 25 percent annual savings compared to Gilenya
- 25 percent annual savings compared to Rebif
- 48 percent annual savings compared to first year of Lemtrada
Future developments in MS therapy
Ocrevus has created a new path of treatments for MS. We’re starting to see additional studies on medications that affect B-cells, as Ocrevus does, but in different ways. These medications could give us additional tools for treating patients.
Future MS therapies may involve the regrowth of myelin, a fatty substance that insulates nerve fibers in the central nervous system. In patients with MS, the immune system strips myelin away and damages the nerves that are left behind.
Exciting research is being done on stem-cell therapies that could actually reset a patient’s immune system. In theory, this would cause the person’s immune system to stop malfunctioning and causing the harmful lesions of MS. And some researchers are working on harnessing the power of robotics to improve the quality of life for MS patients. Think of a partial Iron Man suit – a streamlined exoskeleton that could help these patients move and function more independently.
Our group focuses on the early diagnosis of MS. In most types of medicine, we intervene as early as possible in the disease process. That’s not always the case in MS treatment, and that’s something we want to change. We hope to be able to use some of these innovative therapies to treat patients early in the MS disease process, before they have a chance to become very sick.
One of my most important goals as an MS doctor is to help my patients forget they have MS. Ocrevus makes this possible. Patients won’t have to worry about taking their medication every day. They’ll be able to focus on enjoying life, not managing their MS. That’s a huge advantage, and it’s one I’m excited to be able to share with my patients.