PULSAR: A Paradigm for Safer, More Effective Personalized Radiotherapy
August 8, 2022
Clinical Trials at UT Southwestern
As an academic medical center, UT Southwestern offers clinical trials that give eligible patients access to the newest therapies. Learn more about clinical trials at UT Southwestern.
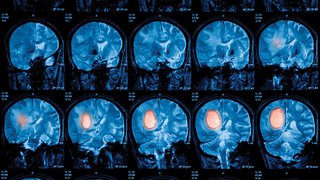
An altogether new approach to radiation therapy that abandons classic daily fractions is now being investigated in rectal, brain, and lung cancer clinical trials.
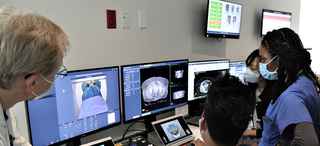
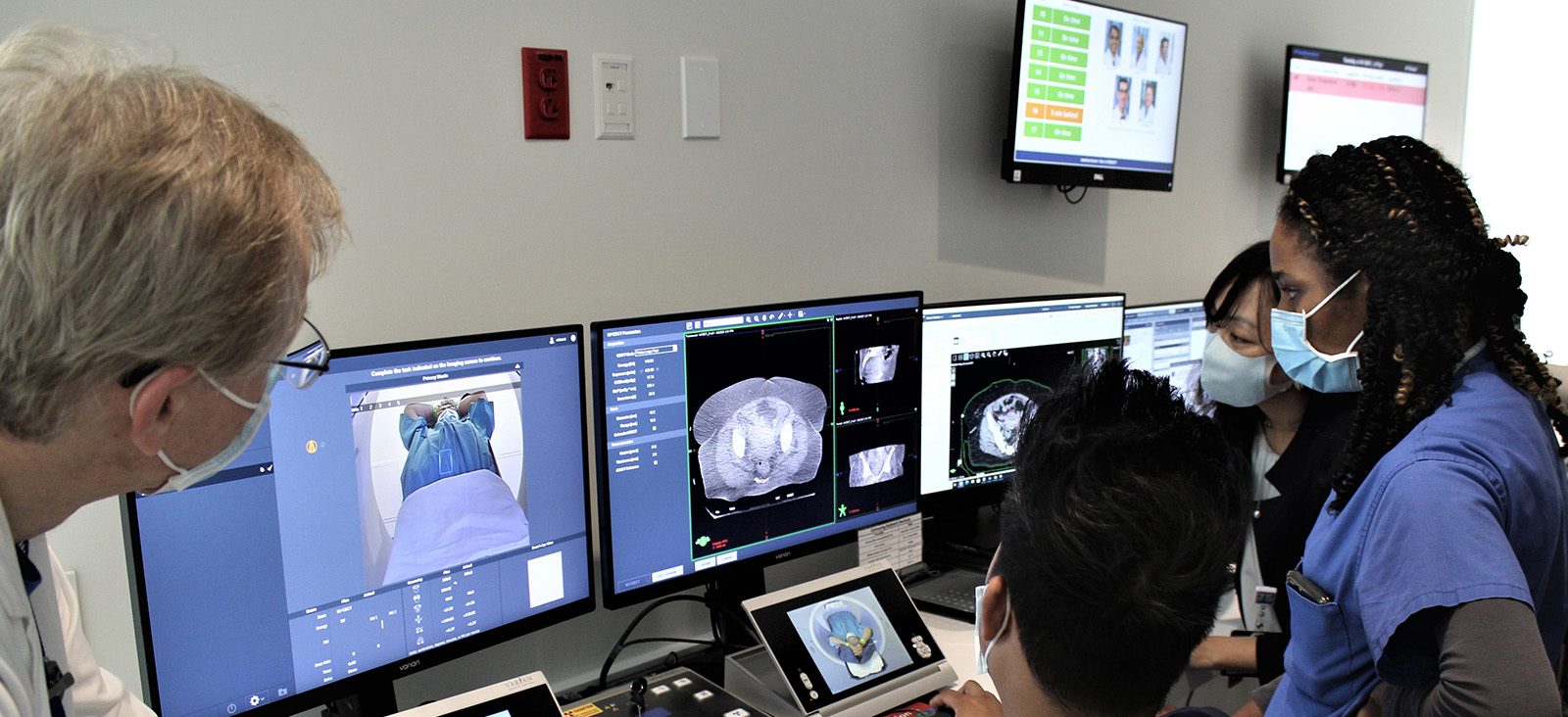
Radiation oncologists at UT Southwestern Harold C. Simmons Comprehensive Cancer Center have pioneered a new technique for delivering more effective and highly personalized radiotherapy. PULSAR®, which stands for personalized ultra-fractionated stereotactic adaptive radiotherapy, could potentially provide benefit above what is currently afforded by strategies such as conventionally fractionated radiation therapy (CFRT).
Robert Timmerman, M.D., is a Professor in the Departments of Radiation Oncology and Neurological Surgery at UT Southwestern and the newly appointed Chair of the Department of Radiation Oncology.
“CFRT has been delivered using typically 30-40 fractions of relatively low doses, given daily over several weeks based on a single dosimetry plan prepared at the start of treatment,” Dr. Timmerman says. “This approach has been used for over 100 years and constitutes a one-size-fits-all care plan.”
Conventional Approaches
Conventional fractionated approaches exploit repair differences between the targeted tumor and nearby normal tissue in reaction to the injury caused by a low daily dose of radiation. The improved repair of small, daily fraction injuries within normal tissue compared with the tumor allows for a therapeutic benefit derived from relative tissue repair biology.
In recent decades, improvements in radiation delivery and imaging have facilitated a new technique that allows for ablative doses, which permit geometric avoidance of surrounding normal tissue and thereby lessen the need to deliver a low daily dose. UT Southwestern pioneered this now popular treatment called stereotactic radiotherapy (SAbR). After more than two decades of experience, the UT Southwestern team plans to take SAbR to the next level, developing a personalized form called PULSAR.
Both CFRT and SAbR treatments are given daily in practice and continue to be referred to as “fractions.”
“We hope our experience leads to more investigation, both observational and mechanistically, of PULSAR dosing schedules for optimum immune preservation and stimulation.”
Robert Timmerman, M.D.
Recent Advances
Recent technological advances have allowed for rapid replanning of a course of radiation therapy in response to patient-specific changes, such as tumor growth or shrinkage. This technique is called adaptive therapy and is a start toward personalized therapy.
Building upon this strategy, PULSAR abandons classic daily fractions entirely and employs a limited number of fairly large dose pulses in the ablative range that are separated by a considerable time, usually weeks or months.
“Separating individual pulses by longer periods of time allows for potentially significant changes to be observed in the tumor tissue, its microenvironment, adjacent tissues, and systemically,” Dr. Timmerman says. “These changes can be analyzed and, if relevant, can prompt adaptive replanning for a more personalized and, hopefully, more effective overall therapy.”
Preclinical Study
Dr. Timmerman and his team performed early exploration of PULSAR alone and in combination with immunotherapy in immune-activated and resistant syngeneic immunocompetent mouse models of cancer.
The results, reported in the International Journal of Radiation Oncology, Biology, Physics, indicated that administering single-agent immune checkpoint blockade (ICB) during or after radiation, and spacing radiation treatments 10 days apart as with PULSAR, achieved better tumor control than traditional daily fractions. The results also indicated that CD8+ depleting antibody abrogated tumor control in the PULSAR combination treatment (meaning the improved effect was related to immunity), and certain treatment schedules induced immunologic memory (indicative of an adaptive immune response).
“To our surprise, we found that by spacing radiation treatments 10 days apart rather than traditional daily fractions, there was a synergistic effect between ICB and the radiation,” Dr. Timmerman says. “By waiting 10 days or longer, the radiation is acting similar to a vaccination followed by a booster shot.”
By allowing more time to evaluate changes for adaptation of an individual patient’s course, the PULSAR technique offers the promise of personalized treatment. Furthermore, completed clinical trials already show PULSAR to be less toxic than other radiation approaches.
Clinical Trials
Based on the encouraging preclinical results, PULSAR is now being investigated in rectal, brain, and lung cancer clinical trials at UT Southwestern. Within each trial, correlative studies are also planned to better understand the effects of PULSAR on immune cells within the tumor microenvironment.
Dr. Timmerman further explains that most preclinical models and new clinical trials are designed with ablative fractions separated by 24 to 48 hours. The recent data might point toward a shortcoming in design when adding immunotherapy to the common daily radiation treatment paradigms.
“We hope our experience leads to more investigation, both observational and mechanistically, of PULSAR dosing schedules for optimum immune preservation and stimulation,” Dr. Timmerman says.
Robert Timmerman, M.D., is a Professor in the Departments of Radiation Oncology and Neurological Surgery at UT Southwestern Medical Center. He serves as Chair of Radiation Oncology.