How Does CAR T-Cell Therapy Work?
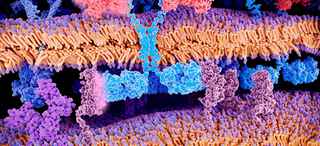
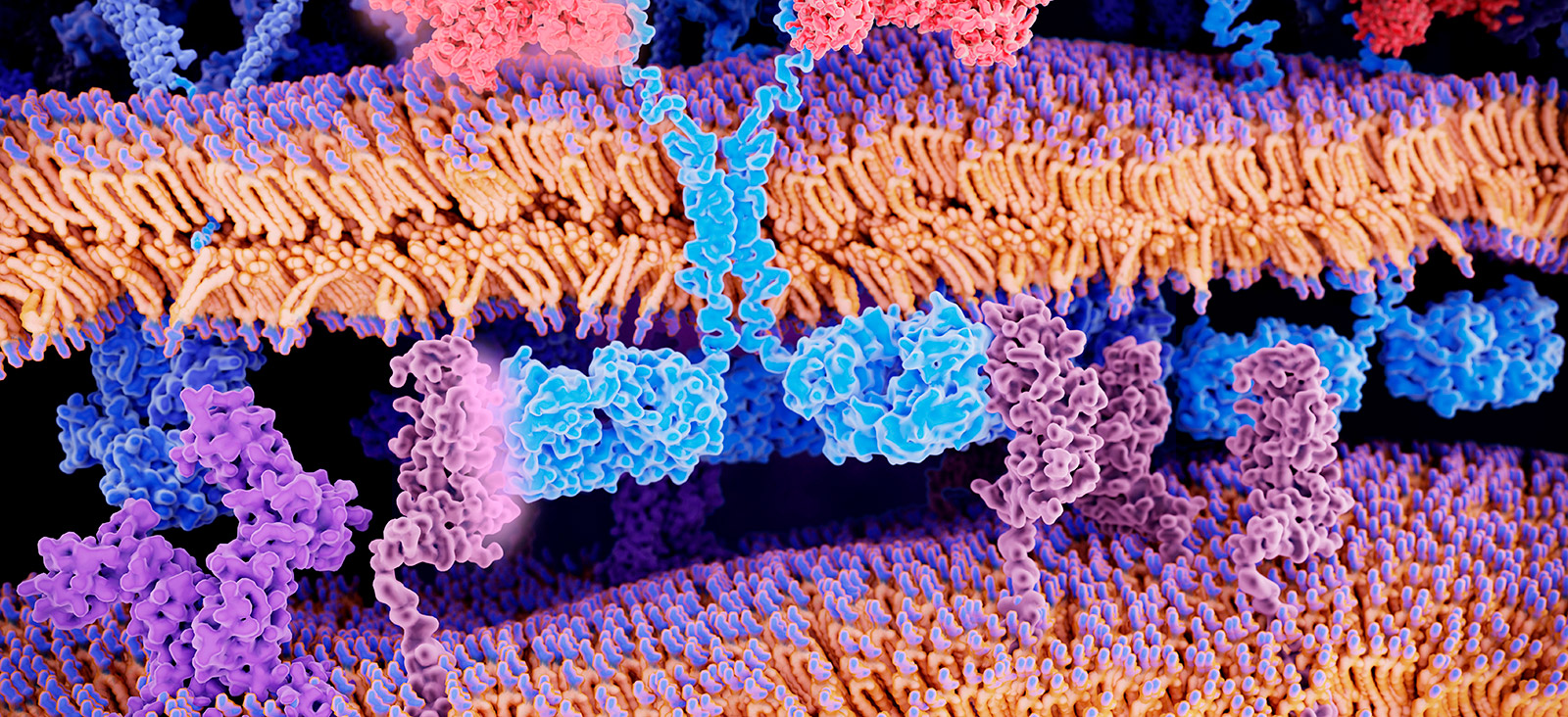
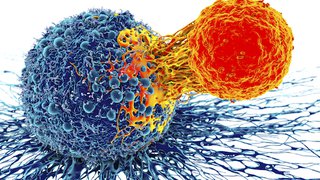
The function of T-cells in the immune system is to destroy abnormal or infected cells, such as viruses and cancer. In some patients, T-cells cannot detect or destroy cancer cells, or there are not enough T-cells. CAR T-cell therapy genetically modifies T-cells so they can better detect, target, and destroy cancer cells.
Our cancer specialists evaluate patients to determine whether they qualify for CAR T-cell therapy. The treatment process, which typically takes 12 to 16 weeks, has several steps:
- Blood collection: Our doctors begin by drawing blood from the patient. The blood is processed to separate some T-cells, and the remaining blood is returned to the patient.
- Genetic alteration of the T-cells: In a special laboratory, doctors genetically modify the T-cells using a disarmed virus. The T-cells produce molecules on their surface known as chimeric antigen receptors (CARs). Doctors then culture the new CAR T-cells so that they multiply into the millions. This process can take several weeks.
- Chemotherapy: The patient receives a brief regimen of chemotherapy to deplete certain immune cells that might attack the new T-cells.
- Infusion: The patient receives the new T-cells in an infusion procedure and typically stays in the hospital for one to two weeks.
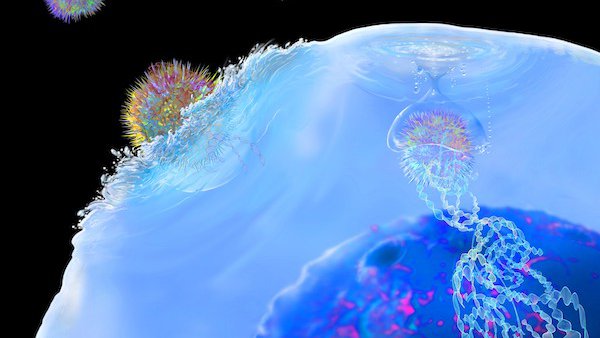
The genetically modified cells continue to multiply in the patient’s body as they circulate through the bloodstream. The new T-cells recognize and attach to a specific antigen (protein) on cancer cells, enabling the T-cells to destroy the cancer cells.
Potential Side Effects of CAR T-Cell Therapy
It is important for patients to receive CAR T-cell therapy from an expert team of cancer specialists at a facility like UT Southwestern. Our cancer doctors have the necessary experience, gained through our participation in clinical trials, to monitor patients for side effects such as:
- Cytokine release syndrome, an inflammatory reaction that produces symptoms ranging from mild and flu-like (fever, nausea, headache) to serious and life-threatening
- Low red blood cell count (anemia)
- Low white blood cell count (neutropenia)