As we pursue new therapies for heart failure, sometimes we stumble upon what the late Bob Ross referred to as “happy little accidents.” Sodium-glucose cotransporter 2 (SGLT2) inhibitor drugs for heart failure are one of my favorite research surprises.
SGLT2 inhibitors were originally designed to treat hyperglycemia, or high blood sugar, in patients with type 2 diabetes. But from 2015-2020, seven trials of four SGLT2 inhibitors were completed with each demonstrating a reduced risk for heart failure hospitalization.
The findings were summarized in a meta-analysis of seven trials that I led and published in 2020. It included nearly 47,000 patients. In those analyses, my colleagues and I demonstrated the remarkably consistent benefits across this class of medications on heart failure outcomes in patients with type 2 diabetes.
These observations led to the design of four more clinical trials: two of patients with heart failure with reduced ejection fraction (HFrEF); and two of patients with heart failure with preserved ejection fraction (HFpEF). The purpose of these studies was to study SGLT2 inhibitors for heart failure treatment, independent of diabetes status.
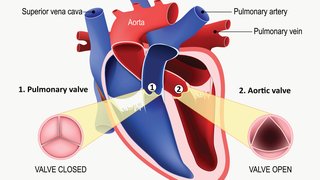
HFrEF is when the heart muscle is not able to contract adequately and, therefore, expels less oxygen-rich blood into the body. HFpEF is when the heart muscle becomes too stiff to pump blood efficiently.
Three of the four trials are complete, and results showed that each of the two SGLT2 inhibitors empagliflozin and dapagliflozin substantially reduced the risk of hospitalization for heart failure.
Clinical trial results suggest that SGLT2 inhibitors can reduce acute decompensated heart failure hospitalizations by 30%, regardless of whether the patient has type 2 diabetes.
But the big question is: Why?
Shifting the focus to 'heart drugs'
It’s well-documented that type 2 diabetes can be a precursor to heart failure. Much of the research surrounding the heart failure benefit of SGLT2 inhibitors has focused on the crossover between heart failure and kidney function and physiology – independent of diabetes and unrelated to the effects of SGLT2 inhibitors on blood sugar.
Diabetes and heart failure are common. Approximately 1 in 5 people will develop heart failure in their lifetime, and an estimated 11% of U.S. adults have type 2 diabetes. Many cardiologists who don’t commonly manage hyperglycemia medications have yet to adopt the use of SGLT2 inhibitors to treat heart failure despite compelling and consistent results from clinical trials. Part of our ongoing challenge in getting the word out about SGLT2 inhibitors for HFrEF will be shifting our vocabulary, calling them “heart drugs” rather than “diabetes drugs.”
Dapagliflozin (brand name Farxiga®) and empagliflozin (Jardiance®) are the first SGLT2 inhibitors to be approved by the U.S. Food and Drug Administration (FDA) for both type 2 diabetes and heart failure risk reduction.
We expect SGLT2 inhibitors to enter the major U.S. heart failure treatment guidelines over the next few years – a significant milestone in connecting more patients to life-improving care.
The first three pillars of heart failure treatment
Prior to SGLT2 inhibitors, we relied on three types of heart failure medications to reduce symptoms, delay or prevent the need for a left-ventricular assist device (LVAD) or a heart transplant, and reduce mortality. All three reduce workload on the heart in various ways, but SGLT2 inhibitors are a true crossover class of drugs, targeting processes in the kidneys rather than the heart.
1. ACE inhibitors, ARBs, and ARNIs
Angiotensin converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARBs) open the blood vessels and lower blood pressure. These changes slow the progression of heart failure by reducing the heart’s workload and helping it pump more efficiently. These most prescribed HFrEF drugs can cause side effects such as headache, diarrhea, dry cough, kidney dysfunction, and increased potassium levels.
The angiotensin receptor-neprilysin inhibitor Entresto® (sacubitril/valsartan) represents a relatively new class of drugs that relaxes the blood vessels and reduces excess body fluid, reducing the heart’s effort to pump blood through the body. Side effects and kidney risks with ARNIs are similar to ACE inhibitors and ARBs. Some guidelines consider all three of these classes of drugs to be at a similar level of safety and effectiveness, though Entresto was proven superior to the ACE inhibitor enalapril in a head-to-head randomized trial.
2. Beta blockers
These medications reduce the strength of heart contractions and slow the heart rate. Over time, beta blockers can slow the progression of heart failure and potentially strengthen the heart muscle. Metoprolol succinate (Toprol XL) and carvedilol (Coreg) are the two most commonly prescribed beta blockers for heart failure. Side effects can include depression, fatigue, and memory problems.
3. Mineralocorticoid antagonists
These drugs are weak diuretics that can help reduce the risk of death and other long-term events related to worsening heart failure. They are used to treat heart failure and high blood pressure by inhibiting aldosterone, a hormone secreted by the adrenal glands. These drugs also decrease sodium reabsorption, increasing the amount of water the kidneys excrete. Side effects can include low blood pressure and higher potassium levels in the blood.
SGLT2 inhibitors: The fourth pillar
The exact reasons why this newest class of drugs treats HFrEF is still being researched. One of the leading theories is that this change is mediated through fixing sodium-related problems in the kidneys, restoring normal kidney physiology. Several hypotheses have been developed about how SGLT2 inhibitors may work to reduce heart failure risk:
- Reduce inflammation: Diabetes and atherosclerosis, as well as heart failure, cause inflammation and vice versa, and SGLT2 inhibitors are thought to decrease inflammation, independent of their effects on blood sugar.
- Restore normal feedback within the kidney: The kidney filters glucose into the urine through a cluster of capillaries at the beginning of the kidney tubule (glomerulus). Then glucose is taken back into the bloodstream (uptake) by SGLT2. Drugs that block SGLT2 also block glucose and sodium re-uptake, which results in the kidney sensing normal sodium and chloride levels in the urine. That reflects appropriate blood pressure and intravascular volume – signaling the kidneys to reduce blood flow and normalize blood pressure within the glomerulus.
- Reduce RAAS system activation: The renin-angiotensin-aldosterone system (RAAS) regulates blood volume and blood pressure, as well as other fluid balances in the body. Subsequently, blood vessel strain is reduced, which decreases blood pressure and requires less work from the heart upstream. When we block RAAS, we reduce nervous system responses that interfere with glucose/sodium uptake. Theoretically, this action also may help restore kidney function and subsequently reduce heart failure complications.
- Reduce sympathetic nervous system activity: This reduces the heart rate and blood pressure because we are essentially turning off the adrenaline nervous system response.
- Reduce intravascular volume: Blocking glucose and sodium re-uptake from the urine causes a diuretic effect (increased urination and excretion of sodium in the urine. In turn, that shrinks ventricle dimensions of the heart, reducing heart muscle wall tension and reducing oxygen demand.
- Increase circulating ketone bodies: SGLT2 inhibitors increase circulating ketones – byproducts of breaking down fat, especially in the fasting state. Ketones are very efficient energy producers for the heart, so in theory, this may decrease oxygen demand and reduce the heart’s workload.
- Increase red blood cell production: SGLT2 inhibitors increase the release of a hormone called erythropoietin from the kidneys. Erythropoietin helps the body make red blood cells, which carry oxygen to the heart and help increase the blood’s oxygen-carrying capacity.
UT Southwestern was a site for the first three clinical trials to demonstrate a direct effect on HFrEF “decongestion” (reduced fluid volumes) and improved clinical outcomes by SGLT2 drugs, specifically dapagliflozin and empagliflozin. Dapagliflozin also has been shown to slow kidney disease progression, independent of diabetes status.
SGLT2 inhibitors cause very few side effects. There is an approximately 5% increased risk of genital yeast infections; in the trials, affected patients typically developed no more than one infection, most were treated with an over-the-counter course of therapy, and most were able to resume study medication once the infection was resolved.
Due to the rare occurrence of diabetic ketoacidosis in patients with type 2 diabetes taking SGLT2 inhibitors, if there is a day in which patients won’t eat as much as normal for any reason, we recommend skipping their daily SGLT2 inhibitor dose. This “sick day” rule is familiar to patients with type 2 diabetes, who know to skip or reduce their short acting insulin and diabetes medicine on low calorie-consumption days.
SGLT2 drugs are expensive. However, robust clinical evidence has resulted in many insurance companies covering at least one of the members of the drug class as a preferred therapy.
The future of SGLT2 inhibitors for heart failure
Several clinical trials are in design or launching to determine whether SGLT2 inhibitors offer other potential cardiac benefits, such as improving the heart muscle function. Additional trials include measuring the effects on broader populations with kidney disease and assessing SGLT2 inhibitor effects after a heart attack in patients who do not have diabetes.

Future research will help cardiologists determine which patients might benefit most from SGLT2 inhibitors and at what point in the disease process.
Over the course of my career, I’ve witnessed the development of numerous classes of diabetes drugs that have now been proven to have cardiovascular safety, such as the thiazolidinediones, various forms of long-acting (basal) insulin, and dipeptidyl peptidase 4 inhibitors, as well as two classes with proven cardiovascular benefits – the SGLT2 inhibitors and the glucagon-like peptide 1 receptor agonists.
Since regulatory agencies in the U.S. and Europe began requiring cardiovascular safety assessment in 2008, I have had the privilege of being involved in the clinical trials assessment of most of the diabetes drugs developed in that time.
Yet, there is always something new to discover and something unexpected that we find. Working closely with our colleagues in endocrinology and nephrology, I have no doubt that more “happy little accidents” will arise from our continued research in the crossover between diabetes, cardiovascular disease, and kidney disease. And if we are fortunate, we might find the key to preventing or curing heart failure altogether.
To visit with a specialist about heart failure treatment options, call 214-645-8000 or request an appointment online.