Search for opportunities to participate in a clinical research study.
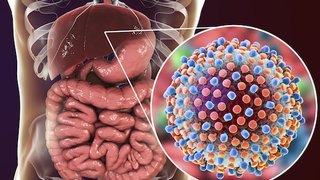
Hepatitis C
New Patient Appointment or 214-645-8300
MedBlog
Results: 5 Locations
Digestive Disease Clinic
at Outpatient Building 1801 Inwood Road, 6th Floor, Suite 102Dallas, Texas 75390 214-645-0595 Directions to Digestive Disease Clinic at Outpatient Building, Dallas Parking Info for Digestive Disease Clinic
Digestive and Liver Diseases
at UT Southwestern Medical Center at Las Colinas 6121 N. State Highway 161, 3rd Floor, Suite 300Irving, Texas 75038 469-647-4705 Directions to Digestive and Liver Diseases at UT Southwestern Medical Center at Las Colinas, Irving
UT Southwestern Digestive and Liver Diseases at Texas Health Dallas
at Professional 4 8440 Walnut Hill Lane, Suite 700Dallas, Texas 75231 214-645-7408 Directions to UT Southwestern Digestive and Liver Diseases at Texas Health Dallas at Professional 4, Dallas
UT Southwestern University Hospital Liver Transplant at El Paso
125 Hague Avenue, Suite 500El Paso, Texas 79902 877-392-1528 Directions to UT Southwestern University Hospital Liver Transplant at El Paso at Oregon Medical Building, El Paso
University Hospital Kidney and Liver Transplant Clinic
at Professional Office Building 2 5939 Harry Hines Blvd., 7th Floor, Suite 700Dallas, Texas 75390 214-645-1919 Directions to University Hospital Kidney and Liver Transplant Clinic at Professional Office Building 2, Dallas Parking Info for University Hospital Kidney and Liver Transplant Clinic