Power within: How a new melanoma treatment shrinks tumors from the inside
April 22, 2026
Sometimes, the most useful strategies to treat cancer start inside the tumor itself. This is proving to be true for advanced melanoma. It’s the third-most common skin cancer and tends to be aggressive, accounting for the most skin cancer-related deaths.
When traditional therapies are not enough to control advanced melanoma, a special type of immune cell living within the tumor called a tumor infiltrating lymphocyte (TIL) can be the key for effective treatment – and a new way to approach solid tumors.
TIL therapy is a personalized form of immunotherapy that dramatically expands the number of a patient’s unique cancer-fighting lymphocytes. These immune cells have identified the cancer as an invader and are attempting to fend it off naturally.
With aggressive cancers like melanoma, the tumor’s spread can easily outpace the body’s ability to create more cancer-eliminating lymphocytes. But TIL therapy gives the immune system a chance to gain the upper hand.
In a laboratory, specialists can duplicate the patient’s unique lymphocytes into the billions, more than the body could make on its own. When the new squad of lymphocytes is infused into the patient, it gives their immune system a better chance of successfully completing its mission.
TIL therapy has been studied for decades with success limited by logistical challenges. Recent advances in cell manufacturing and preservation are now leading to the first cellular therapies approved for use in solid tumors, including melanoma.
UT Southwestern’s Harold C. Simmons Comprehensive Cancer Center is an authorized TIL treatment center. Our care team combines the knowledge of multiple experts in advanced melanoma care, surgery, and cellular therapy. These specialists deliver precise, personalized treatment for patients whose melanoma has progressed despite standard therapies.
“Basically, we’re incubating the T cells and activating them with different cytokines to grow them into bigger batches over a span of weeks. The timeline is an important consideration in the treatment planning process.”
Daniel Wang, M.D.Associate Professor of Internal Medicine in the Division of Hematology and Oncology
How does TIL therapy work for melanoma?
TIL therapy for advanced melanoma, available as lifileucel, is the first treatment developed specifically for patients with unresectable or metastatic melanoma that worsened after immunotherapy or targeted therapy.
Similar to CAR T-cell immunotherapy, TIL uses a patient’s own immune cells as the foundation for treatment. While CAR T‑cell immunotherapy involves genetically modifying a patient’s immune cells, TIL therapy maximizes the production of their naturally occurring lymphocytes (a combination of T cells and B cells).
TIL therapy is not a single appointment or given in cycles. It is a carefully coordinated, multistep process that involves collaboration across multiple specialties.
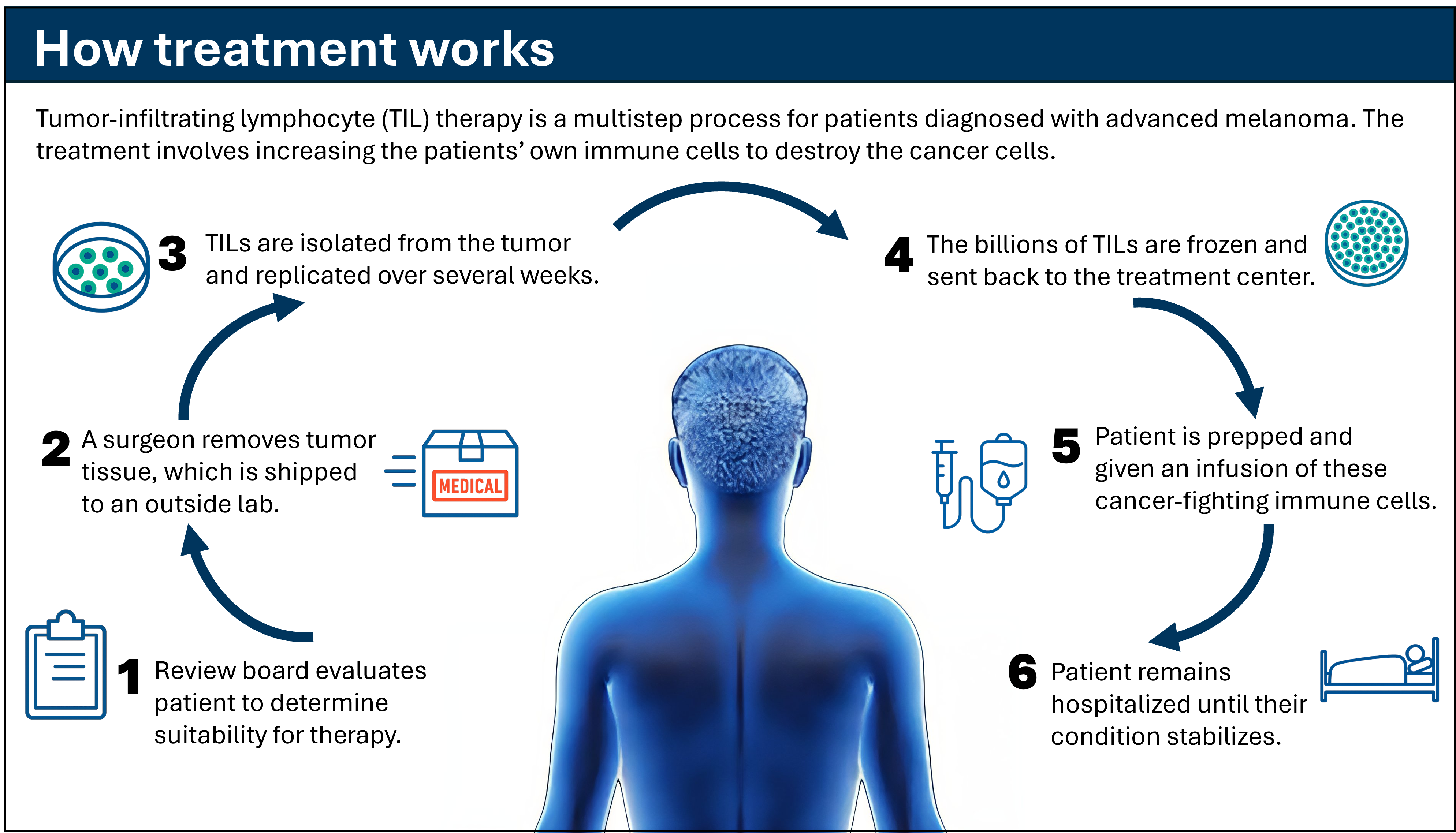
Step 1: Patient evaluation and tumor board review
We begin with a thorough evaluation to determine whether TIL therapy is appropriate. Because the treatment is intensive, patients must be healthy enough to have surgery, chemotherapy, and a special type of cytokine therapy, known as interleukin-2 (IL-2).
A key requirement is the presence of a tumor that can be safely removed to obtain the TILs. Each case is reviewed by a multidisciplinary tumor board that includes medical oncologists, surgeons, and cellular therapy specialists. This collaborative approach helps ensure that patients are selected thoughtfully and that treatment planning is individualized.
Step 2: Tumor tissue sampling
Once approved, a surgeon removes a portion of the tumor, selecting a site that provides enough tissue for immune cell extraction while minimizing risk.
Step 3: Cell extraction and replication
The tumor tissue is shipped to a cell therapy laboratory, where scientists isolate the TILs. These cells are then placed in a controlled environment that encourages them to grow and multiply using immune‑stimulating signals such as interleukin‑2 (IL‑2).
Over several weeks, millions to billions of tumor‑recognizing lymphocytes are produced.
Advances in manufacturing have significantly shortened this phase – what once took about six weeks can now often be completed in three to four weeks.
TIL therapy is designed as a one-time treatment. Unlike many cancer therapies that require ongoing cycles over months or years, TIL involves a single infusion followed by monitoring.
Step 4: Freezing and return shipment
One of the most important technological breakthroughs in modern TIL therapy is the ability to freeze the expanded cells without compromising their function. This allows the therapy to be produced centrally and transported back to the treating cancer center, eliminating the need for patients to travel long distances for care.
Step 5: Preparation and infusion
Before receiving the TIL infusion, patients get lympho-depleting chemotherapy. This step temporarily reduces their existing immune cells, creating space for the incoming TIL cells to do their job more effectively. The high-volume lymphocytes are infused as a one‑time treatment. Shortly afterward, patients get an IL‑2 infusion to help the new cells thrive.
Step 6: ICU monitoring and recovery
In contrast to some other immunotherapies, TIL therapy does not appear to cause the same pattern of long-term immune-related side effects, such as chronic inflammation of the thyroid, lungs, or intestines.
However, the IL-2 infusion can cause significant, yet manageable, side effects including increased risk of infection, gastrointestinal distress, flu-like symptoms, fatigue, and skin rashes. High-dose IL-2 can cause capillary leak syndrome, in which fluid shifts out of blood vessels into surrounding tissues. This can lead to low blood pressure, swelling, organ dysfunction, and strain on the heart and lungs.
Because of these risks, careful patient selection is critical. People considering TIL therapy should discuss their overall health, prior treatments, and goals of care with their oncology team.
“This is a therapy we like to start thinking about earlier rather than later. We need patients to be pretty health to withstand the rigors of treatments. We don’t want to miss the opportunity to start patients on something that could be really groundbreaking for them,” said Daniel Wang, M.D., Associate Professor of Internal Medicine in the Division of Hematology and Oncology and Leader of the Sarcoma/Melanoma Disease Oriented Team at Simmons Cancer Center.
After the infusions, patients are monitored in the intensive care unit until their blood count and overall condition stabilize. Most treatment-related effects gradually improve during their hospital stay.
Related: Read “Immunotherapy and the marathon against melanoma: Terry's story”
What outcomes should a patient expect?

In the clinical trial that led to Food and Drug Administration (FDA) approval, about one-third of patients with advanced melanoma experienced a measurable reduction in tumor size after a single infusion of lifileucel. A smaller percentage saw their tumors disappear completely on imaging.
Many of these responses were long-lasting. Among patients whose tumors shrank, a substantial proportion remained free of disease progression for a year or more after treatment. Some responses have lasted several years. Emerging data suggest that giving TIL therapy in earlier lines of treatment may lead to better outcomes.
Outcomes with immunotherapies for melanoma vary based on an individual’s overall health and the nature of their cancer. Researchers continue to study which patients are most likely to benefit.
“TIL therapy takes a village, and that’s what UT Southwestern brings to the table. We have engaged teams, and we have multiple experts in different areas. Surrounding the patient with a multidisciplinary team allows us to successfully offer novel therapies and deliver quality care.”
Sanjay Chandrasekaran, M.D.Assistant Professor of Internal Medicine in the Division of Hematology and Oncology
History of TIL
Research into using TIL for melanoma began in the 1980s, when early studies demonstrated that tumor‑infiltrating immune cells could be manufactured and reinfused to generate meaningful responses in melanoma. For many years, however, the approach was difficult to deliver outside of highly specialized academic centers because of its complexity and months-long manufacturing process.
Researchers at the National Cancer Institute partnered with scientists at Iovance Biotherapeutics to streamline and optimize cell culture techniques, freezing methods, and centralized production.
With the production timeline shortened to just a few weeks, the cells can now be frozen, safely shipped back to the treatment institution, and thawed for infusion without losing their activity. The result is a personalized cell therapy created from a patient’s own immune system.
"As cell therapy has advanced, we’ve been able to expand what we offer patients with solid tumors here at UT Southwestern. TIL therapy is a natural next step for a team that already delivers sophisticated, personalized cancer care and works across disciplines to manage highly complex treatments."
Amena Shahabuddin, MPAS, PA-C, Physician Assistant
A team effort for complex care
Success with TIL therapy requires an all-hands-on-deck approach. At UT Southwestern, this means deep collaboration among melanoma experts and cellular therapy specialist teams. By bringing together surgical experts, dedicated nurses, and cell therapy physicians, we can provide the high level of coordination needed for this logistically complex treatment.
As TIL therapy technology becomes more widely available, it offers a new path for patients whose melanoma has progressed despite other treatments. Our goal is to provide more patients living with advanced melanoma access to this potentially lifesaving personalized immunotherapy.
Meet the team
- Daniel Wang, M.D., is Associate Professor of Internal Medicine in the Division of Hematology and Oncology and Leader of the Sarcoma/Melanoma Disease Oriented Team at Simmons Cancer Center. He specializes in melanoma, advanced skin cancers, and soft tissue and bone sarcomas.
- Sanjay Chandrasekaran, M.D., is Assistant Professor of Internal Medicine in the Division of Hematology and Oncology and specializes in melanoma, advanced skin cancer, and soft tissue and bone sarcomas.
- Amena Shahabuddin, M.P.A.S., PA-C, is a board-certified physician assistant in the Simmons Cancer Center with a focus on cutaneous malignancies and sarcoma.
- Amanda Dann, M.D., is Assistant Professor of Surgery and specializes in surgical oncology for melanoma, non-melanoma skin cancers, and sarcoma.
- Adrienne Shannon, M.D., is Assistant Professor of Surgery and specializes in cutaneous surgical oncology and soft tissue tumors.
- Farrukh Awan, M.D., is Professor of Internal Medicine in the Division of Hematology and Oncology and specializes in bone marrow transplantation and treatments for leukemia and lymphoma.
- Praveen Ramakrishnan, M.D., is Assistant Professor of Internal Medicine in the Division of Hematology and Oncology. He specializes in treating lymphoma and other hematologic malignancies.
To talk with an expert about TIL therapy or melanoma care, make an appointment by calling 214-645-4673 or request an appointment online.













